  
I prefer to automate as much as possible, both because I don’t want the hassle and I want to avoid the potential bias that comes with manual changes. These can be removed manually or you can use a threshold (lower and higher, outside of the physiological range). Remove any gaps. There may be gaps in the data file where the probe had to be shifted for gel application or similar.While smoothing helps getting rid of some of these issues, I prefer to smooth as little as possible. Smooth the data. No data is perfect, and TCD data can be quite noisy for a number of reasons.This helps figuring out the quality, and making sure I’ve measured what I think I have measured. I always look at my data before I start working on it. Read the data file in MATLAB (or similar), then follow these processing steps.This will depend on the software, and I have found it easiest to work with machines that allow direct (during data collection) connection with a computer, feeding into Spike, LabChart or similar programs, as I know how to export data from such software without any fuss. I have used custom Matlab scripts to analyse these, using quite simple steps: In my research, I analyse beat-by-beat blood flow velocity in healthy participants, looking how this changes over time. The custom software may not always be what we are looking for, and diagnosis may not always be the end-game. For research purposes, however, things are a bit different. Usually, it requires training and experience to interpret ultrasound results for diagnostic or healthcare purposes, even with custom software. Now what? How do we analyse this? Some ultrasound machines have their own analysis software that can be used to determine whether the blood flow is normal. 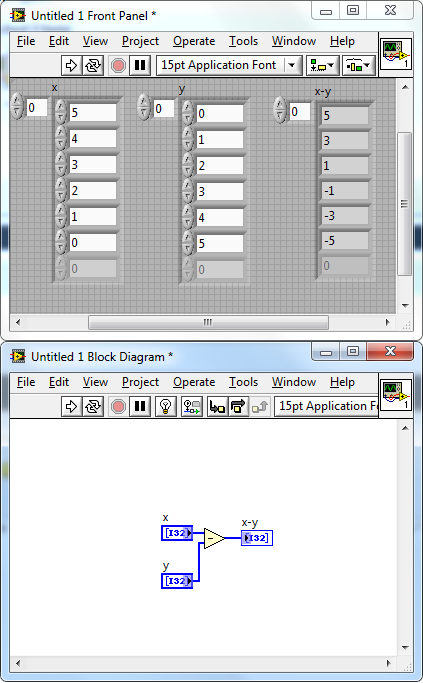
If necessary, an additional channel was set up to calcualte a linear correction for the O2 data if minor probe drift was detectable.Ī few additional columns on the data pad in LabChart Reader were set up to record information included start and end times of selected data segments and number of data points included in slope calculations.In a previous post I described how to use ultrasound to measure brain blood flow. Channel 5 was set up in the Data pad to record average slope (Under the "Slope" > "Average slope" for each selected segment of data (data for each negative slope were manually highlighted in LabChart Reader (highlighting all channels simultanesouly) to encompass as much ~linear negative slope during each closed phase as possible, based on visual estimation of the negative linear portions of the O2 data). The "Times in First Column" option was UNCHECKED and the default sampling rate was added as 0.1 Hz.Ĭhannels 1 and 2 in LabChart Reader were left as is, Channel 3 converted Channel 2 (PO2 in % air saturation) to PO2 in Torr, Channel 4 converted PO2 in Torr to micromoles of oxygen per Liter based on the volume of the respirometer less the volume of the fish (i.e., mass of the animal, assuming 1 mL = 1 g), the reported barometric pressure during the respirometry trial from the nearest weather station (YVR airport), and a solubility coefficient of 1.7206 micromoles O2 per Liter water per Torr atmospheric pressure (see. 
txt file was read in using the "LabChart Text File" option and selecting the appropriate file. txt file for use in LabChart Reader v8.1.9. The "Time elapsed in minutes" data column and "Oxygen" data column from this excel sheet were then saved in a separate. csv respirometry files recorded by NeoFox Viewer v2.4 were separately pasted into the 1st and 2nd column, respectively, of this excel sheet. The "Time" and "Oxygen" data columns from each raw. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
